Publications internes
 extracellular_vesicle
extracellular_vesicle
VIDEODROP: For a fast characterization of Extracellular Vesicles following a standard protocol
The recent interest growth in Extracellular Vesicles (EVs) is governed by the potential that these cell-derived membranous nanoparticles present in terms of theranostic effect.
 lipid_nanoparticle
lipid_nanoparticle
TAMARA: Development of a Simplified Microfluidic Nanoparticle Formulation System Tailored for Preclinical Stages
Developing nanotherapies faces significant formulation challenges, as seen with RNA-LNP-based therapies, which account for over 500 formulations in development, yet only four are FDA-approved.
 extracellular_vesicle
extracellular_vesicle
Quantification of bacteriophages, extracellular vesicles and bacteria in Human stools
It is now largely accepted that the intestinal microbiota plays a key role in Intestinal Bowel Diseases (IBD). There are indications that phages, the viruses infecting bacteria, but also extracellular vesicles (EVs), might play a part in this family of diseases.
 extracellular_vesicle
extracellular_vesicle
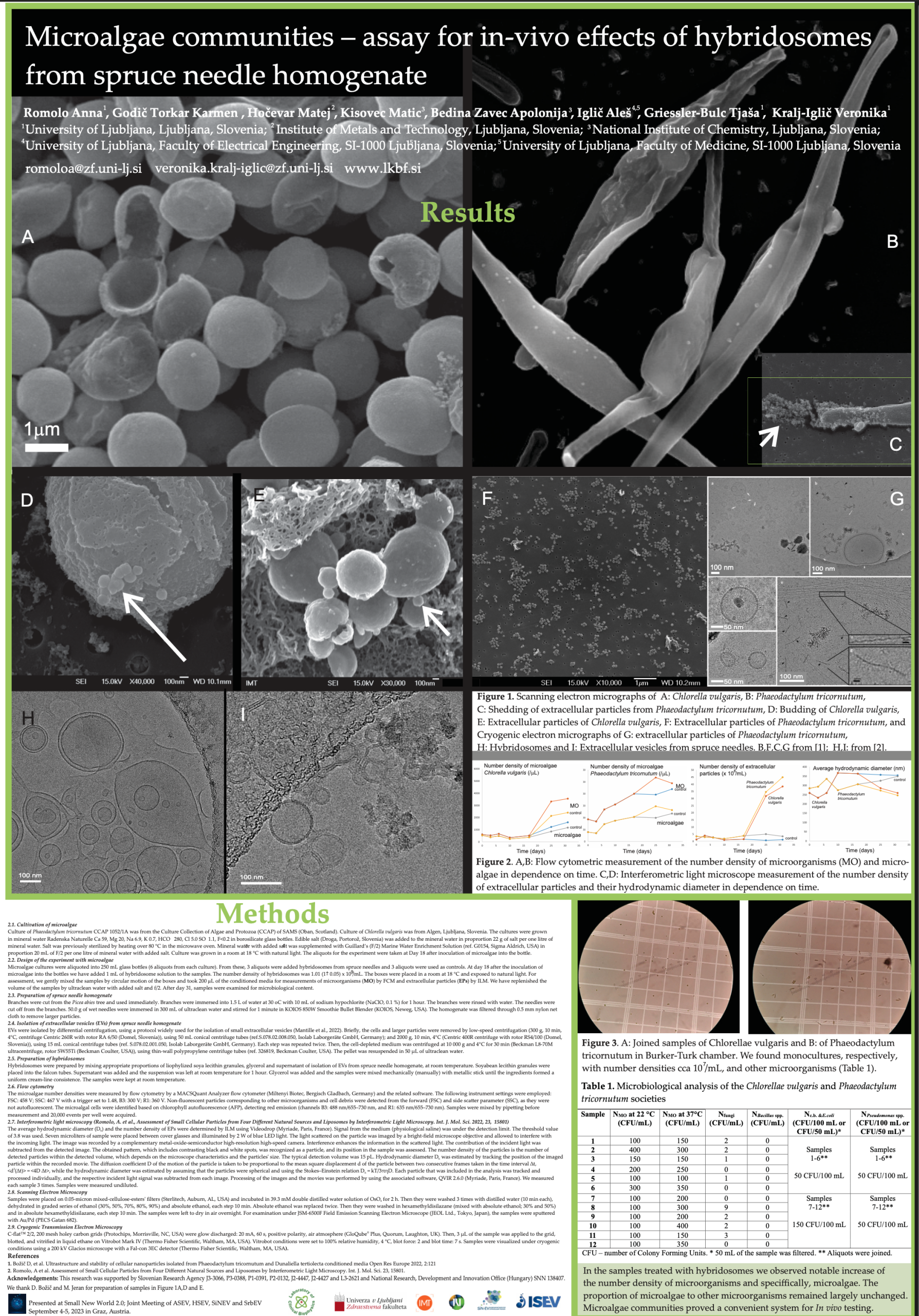
Microalgae communities – assay for in-vivo effects of hybridosomes from spruce needle homogenate
 lentiviral_vector
lentiviral_vector
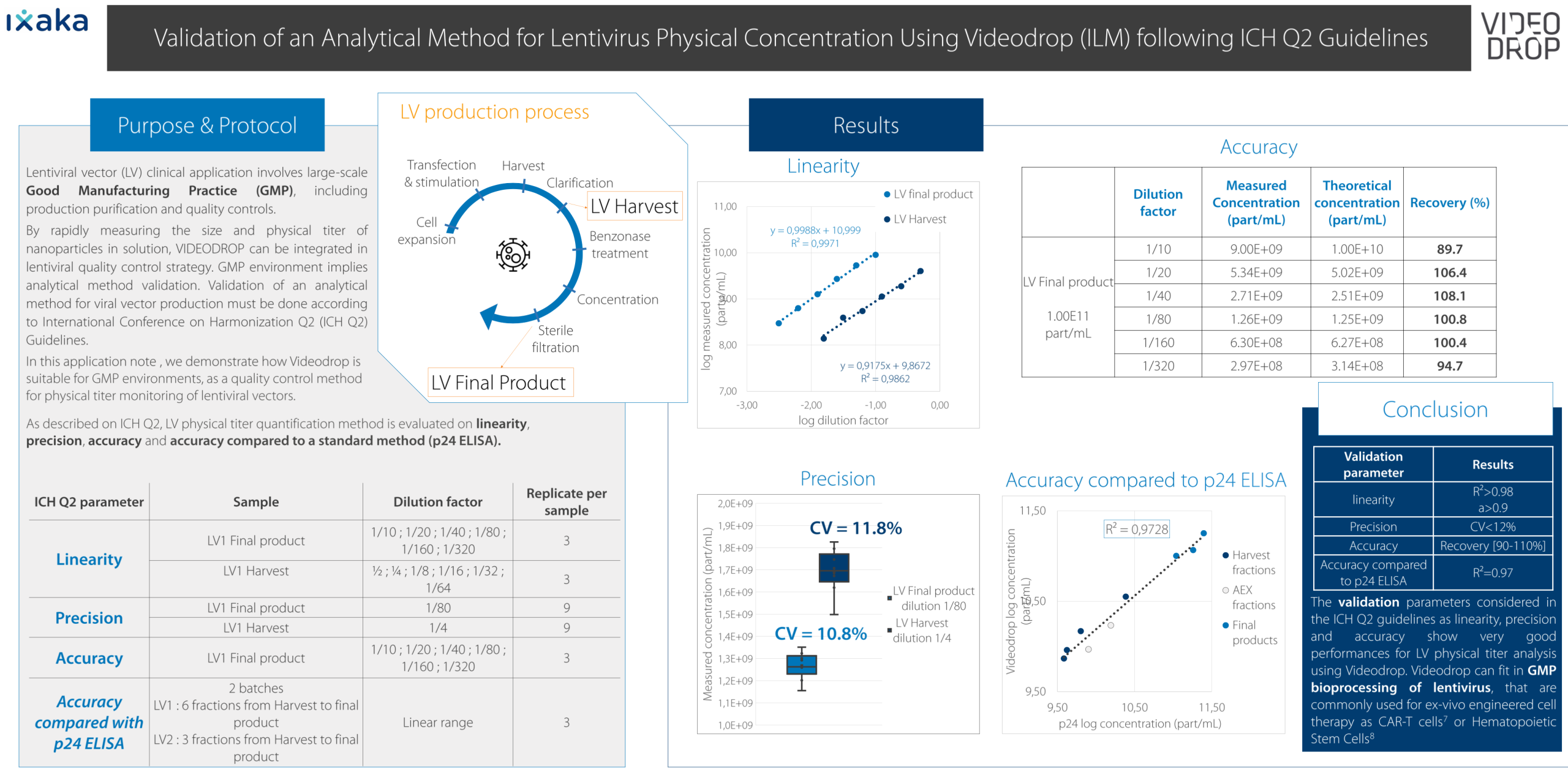
Validation of an Analytical Method for Lentivirus
Lentiviral vector (LV) clinical application involves large-scale Good Manufacturing Practice (GMP), including production purification and quality controls.
 lipid_nanoparticle
lipid_nanoparticle
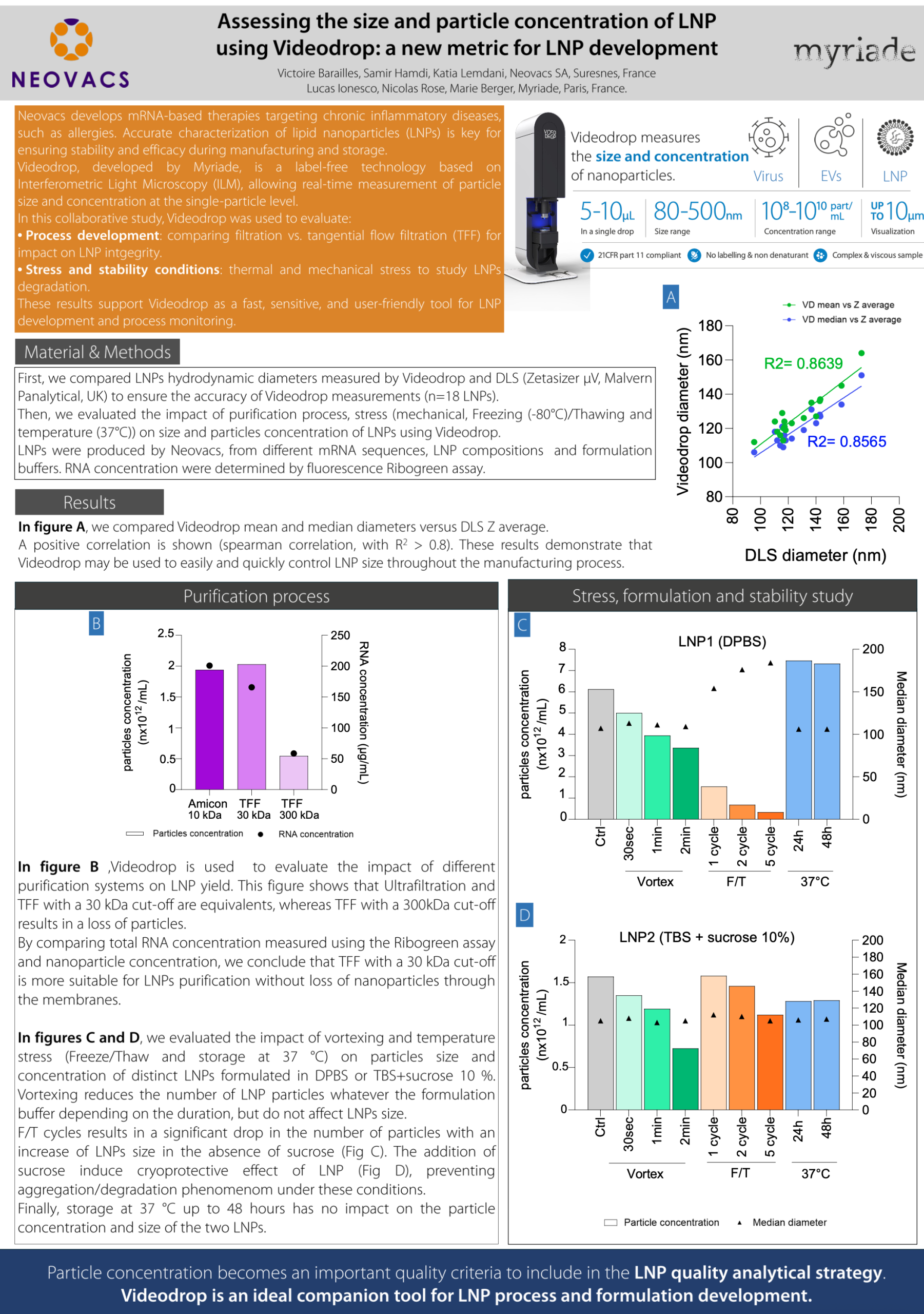
Assessing the size and particle concentration of LNP using Videodrop: a new metric for LNP development
Neovacs develops mRNA-based therapies targeting chronic inflammatory diseases, such as allergies. Accurate characterization of lipid nanoparticles (LNPs) is key for ensuring stability and efficacy during manufacturing and storage
 extracellular_vesicle
extracellular_vesicle
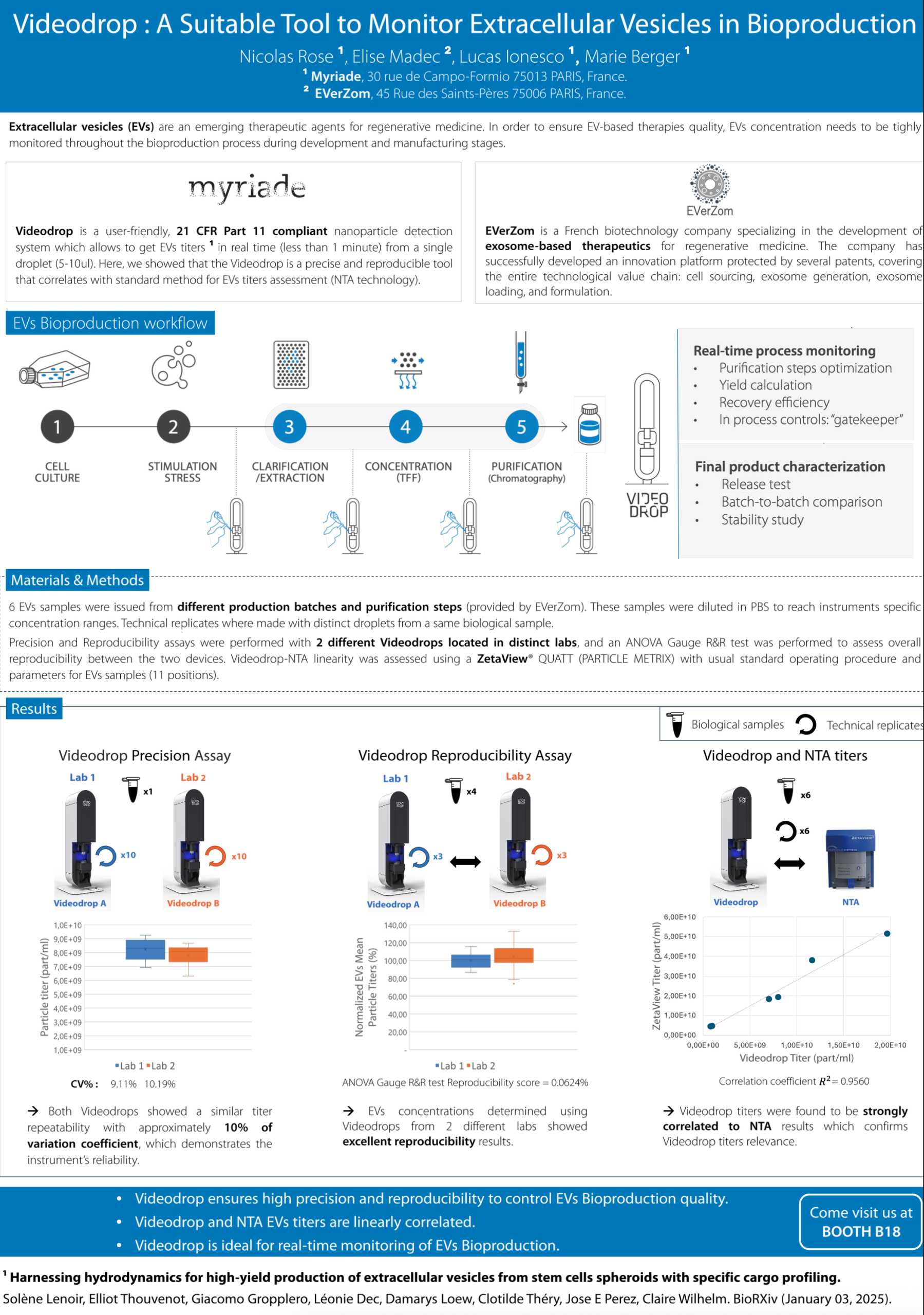
Videodrop: A Suitable Tool to Monitor Extracellular Vesicles in Bioproduction
Extracellular vesicles (EVs) are an emerging therapeutic agents for regenerative medicine. In order to ensure EV-based therapies quality, EVs concentration needs to be tightly monitored throughout the bioproduction process during development and manufacturing stages.
 other
other
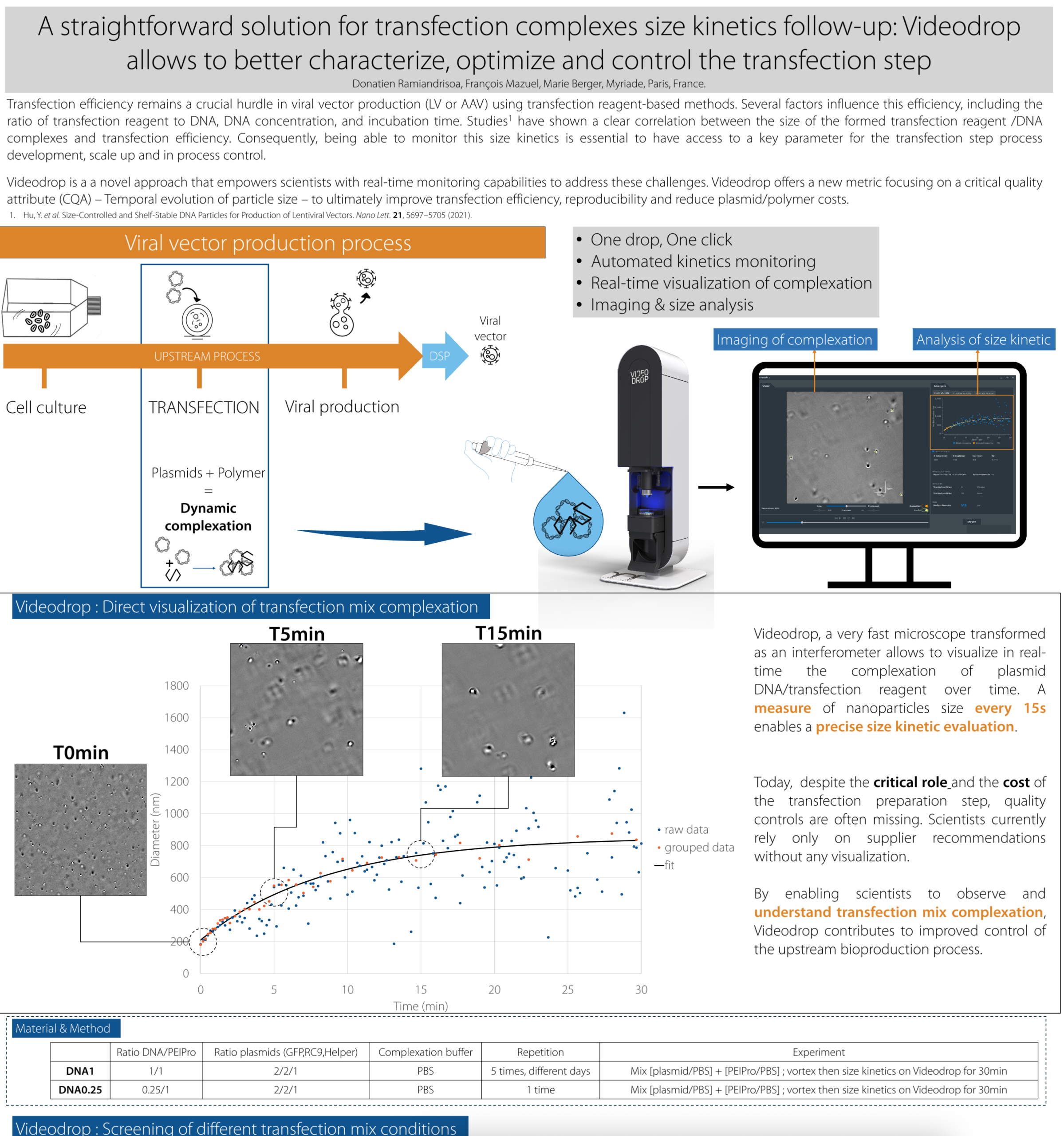
A straightforward solution for transfection complexes size kinetics follow-up
Transfection efficiency remains a crucial hurdle in viral vector production (LV or AAV) using transfection reagent-based methods.
 lipid_nanoparticle
lipid_nanoparticle
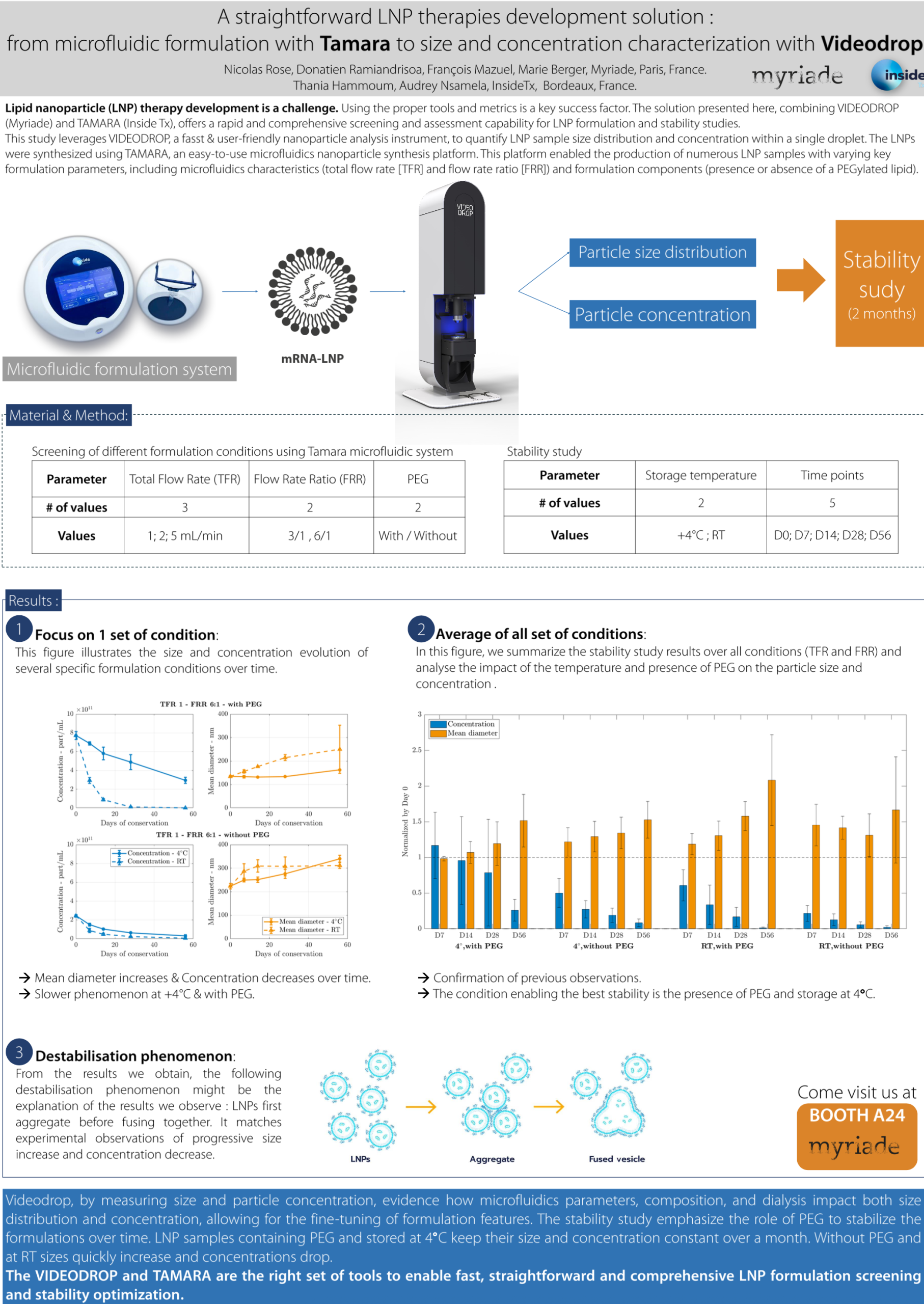
A straightforward LNP therapies development solution: from microfluidic formulation with Tamara to size and concentration characterization with Videodrop
Lipid nanoparticle (LNP) therapy development is a challenge. Using the proper tools and metrics is a key success factor.
Publications externes
Pan et al. [2025]
Mozafari et al. [2025]
Lenoir et al. [2025]
Cavaleri et al. [2025]
Yuyama K. et al. [2025]
Spasovski V. et al. [2024]
Richard M. et al. [2024]
Korenjak B. et al. [2024]
Delehedde C. et al. [2024]
Boulestreau J. et al [2024]
Alexandre L. et al. [2024]
Petrovic M. et al. [2024]
Lapras B. et al. [2024]
Takada et al. [2023]
Troha V. et al. [2023]
André-Grégoire G. et al. [2023]
Poret A.et al. [2022]
Romolo A. et al. [2022]
Andre-Gregoire G. et al. [2022]
Kato R. et al. [2022]
Turkki P. et al. [2021]
Sabbagh L. et al. [2021]
